PDF Version: Notes on the Drude Model
Assumptions of the Drude Model
- The Drude model provides a classical mechanics approach to describing conductivity in metals. This model makes several key assumptions (some of which are better approximations than others).
- Electrons in a metal behave much like particles in an ideal gas (no Coulombic interaction and no collisions between particles). This is called the independent electron approximation.
- Positive charges are located on immobile ions. The electrons do not experience coulombic interaction with the ions, but they do collide with the ions and can change direction and velocity.
- Electrons reach thermal equilibrium by collisions with the ions. Their mean kinetic energy within the lattice at equilibrium is given below. The mass of an electron is represented as me, the average velocity at a given temperature is vT, the Boltzmann constant is kB, and the temperature in Kelvin is T. At room temperature, vT is about 105 m/s.
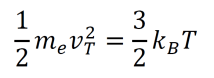
- The average distance of an electron’s free movement between collisions is called the mean free path λ. For metals, the mean free path is typically estimated as 1 nm based on known ionic packing parameters. To calculate the mean time τ between collisions (called the relaxation time), the equation τ=λ/vT is used.
Applying the Drude Model
- To apply the Drude model, the density of the “gas” formed by the free electrons must be known. This parameter is called the conduction electron density n (the number of free electrons per volume).
- The conduction electron density is computed by assuming that each atom contributes ZV conducting electrons. ZV represents the number of outer shell electrons for metal atoms in the ionic lattice. For instance, alkaline Earth metals have a ZV value of 2. Given the density ρm in kg/m3 and the atomic mass M in kg per atom, the conduction electron density is ZVρm/M.
- Given the average time for a collision-free drift τ, the average drift velocity of an electron in a metal can be computed using the equation below. Here, e represents the charge of an electron in Coulombs and ℇ is electromotive force in volts (a vector quantity).
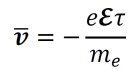
- The number of electrons passing through a given area per unit time Je and the amount of charge passing through a given area per unit time Jc are given below. Here, n is the number of electrons.
![]()
![]()
- The current density j is computed by the equation below.
![]()
- This can be used to derive the following expression, which is equivalent to Ohm’s law V=IR.
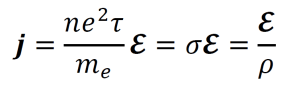
- As such, the conductivity σ and the resistivity ρ are given by the equations below.
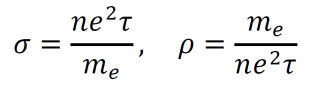
- The mobility μ of an electron in a lattice is given below. The mobility can be interpreted as the ratio of the drift velocity to the applied electric field.

- Using mobility, conductivity and resistivity can be computed by alternative formulas.
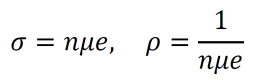
The Drude Model and the Hall Effect
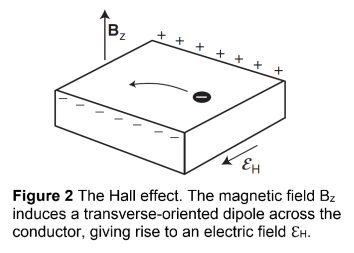
- The Drude model explains the Hall effect, a phenomenon in which an electric field ℇH arises perpendicular to both the current density jx (which points in the direction of electron movement) and the magnetic field Bz.
- The Hall effect occurs when a current flows through a conductor while under a magnetic field. As a result of the magnetic field, positive charges accumulate on one side of the conductor (and negative charges on the other side).
- For electrons to pass through the given region, the field ℇH must cancel the Lorenz force which acts in the opposite direction (the Lorenz force is the sum FL=qE+qv×B of the magnetic force and electric force on a moving charged particle). The magnitude of the Hall field is given below. RH is called the Hall coefficient and can be measured experimentally.
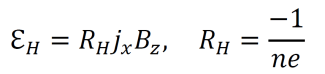
- As the value of RH approaches one, the field ℇH more exactly cancels the Lorenz force. The value of RH varies between types of metals, so some metals are better conductors than others.
The Drude Model and Optical Reflectivity of Metals
- Light can be described as an electromagnetic wave in the form of a transverse plane.
- The electric field for light in the propagating in the z direction is given by the equation below. ℇ0 is the amplitude in the xy plane, λ0 is the wavelength in a vacuum, n is the index of refraction, and κ is a parameter that accounts for attenuation of the light’s intensity inside of a given material.
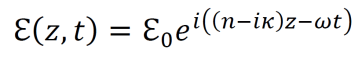
- Alternatively, the equation for the electric field of light propagating in the z direction can be expressed using the dielectric constant of the given material.
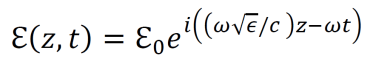
- An electron within the electric field from an electromagnetic wave moves according to the following equation of motion (derived from F=ma). Note that the charge –e and the exponential are distinct.
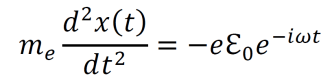
- Solving the differential equation above gives the following result.
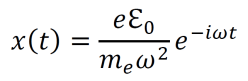
- As a result of the electric field from the electromagnetic wave, the electron undergoes positional displacement in a periodic manner. This leads to a changing dipole moment given by –ex(t).
- Dielectric functions describe the permittivity of given media over time. Permittivity is the amount of charge needed to generate a single unit of electric flux within the medium. Using the above results and some other known formulas, the dielectric function for a material can be derived.
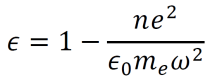
- By setting a parameter ωP2 equal to ne2/meε0, the dielectric function can be rewritten as below. Note that ωP is called the plasma frequency.
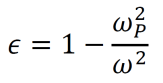
- For ω<ωP, the value of ε is real and negative, making the square root of ε purely imaginary. As such, the light does not transmit into the metal in this case. Since energy is conserved, the light is reflected instead.
- For ω>ωP, the value of ε is real and positive. As such, the light does propagate into the metal in this case.
- Metals reflect low-frequency light and are transparent for high-frequency light. The transition occurs at the plasma frequency ωP.
- The plasma frequency can be measured experimentally or calculated using the conduction electron density n.
The Drude Model and the Wiedemann-Franz Law
- Thermal conductivity is defined by the equation below where Q is the amount of heat transferred per time t, k is the thermal conductivity constant for a given material, A is the cross-sectional area, d is the thickness of the material, and ΔT is the difference in temperature across the material. Note that this definition only describes the simple 1-dimensional case, but analogous formulas can be used for more general situations.
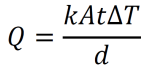
- The Wiedemann-Franz law states that, for any metal at a given temperature, the ratio of thermal conductivity κ to electrical conductivity σ is a constant L. Furthermore, L is proportional to temperature as temperature varies. L is called the Lorenz number.
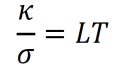
- The ideal gas-related equations from the Drude model can be used to generate an equivalent formula for LT. Despite some minor inconsistencies with experimental data, this theoretical calculation often gives strikingly accurate results.
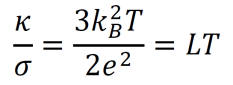
Shortcomings of the Drude Model
- The Drude model does not take into account collisions between electrons themselves. It also does not consider electrostatic interactions between the electrons and the lattice ions.
- The de Broglie wavelengths of electrons with some thermal energy are on the nanometer scale. This means that electrons cannot be treated as classical particles (since they have substantial wave character) under the conditions of the Drude model.
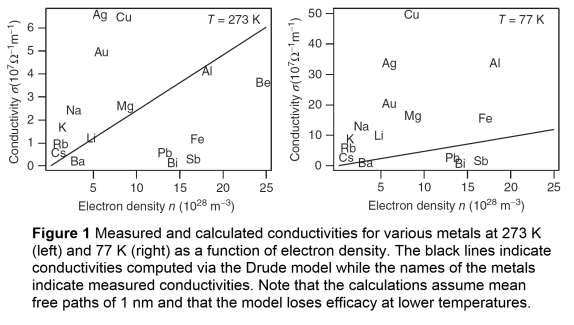
- As mentioned, the Drude model underestimates conductivity of metals at low temperatures. This is because the assumption of a constant mean free path (based on atomic spacing) is incorrect. The mean free path varies greatly with temperature, particularly in pure crystalline substances.
- The Drude model cannot explain the conductivity of alloys. Even small impurities can drastically decrease the conductivity of metals in a way which is not predicted by the Drude model.
- From a classical mechanics perspective, the electrons should contribute greatly to the heat capacity of metals. But this result does not agree with experimental data (and using quantum mechanical models instead resolves the issue).
Reference: Hofmann, P. (2015). Solid State Physics: An Introduction. Wiley.
Hey Logan,
really nice writeup about the Drude model in a more solid-state sense!
As you wrote: the Drude model is just a phenomenological model to explain certain, say, frequency ranges. Going beyond that I would say the most important aspect is the description of conductivity in the visible spectrum. Then it is really useful to find analytic resonances for gold and silver particles. I have wrote about it here from an electrodynamics perspective: https://photonics101.com/light-matter-interactions/drude-model-metal-permittivity-conductivity , maybe you like it as well 🙂
Take care
Robert
LikeLike
Dear Logan,
thank you for your writeup about the Drude model coming from a solid state perspective (I hope my comment is not duplicated…) Phenomenologically the model has its best feats to describe metals in the visible spectrum. Then you can analyze more geometric resonances of particles such as spheres or ellipsoides. Maybe you like my writeup about an electrodynamic approach to the theory on: https://photonics101.com/light-matter-interactions/drude-model-metal-permittivity-conductivity 🙂 – directly starting from a harmonic oscillator (as always 😉
Take care
Robert
LikeLiked by 1 person
Thank you, I’m checking out your site and it looks great!
LikeLike